I was recently interviewed for one of my favourite magazines – Digital FilmMaker! Here is the original with some of my photos from my ‘A Lion’s Tale’ shoot in Kenya; exactly a year ago. Hope you enjoy it and feel inspired to do your own!

- Why will this project be of interest to readers of Digital FilmMaker?
Hopefully this will be of interest to anyone wanting to shoot their first short film and have only just begun to delve into this creative, thrilling world of visual storytelling. Also, that it is indeed possible to do on a very small budget, whilst travelling to amazing places in the process! Natural history differs to drama in that you cannot predict what the wildlife ‘characters’ will do; or control a great deal of external environmental factors. However, with careful planning and preparation during the pre-production stage; it is possible to make an emotive and personal human-wildlife story that resonates with your target audience.
- What had you done project wise in the lead up to this?
This was one of the first and biggest project that I was completely involved in. I studied Zoology during my undergraduate years and played around with cameras in different university societies; but nothing on this scale. I lived in southern Spain all my life before moving to the UK to study as an 18-year old, and upon graduating I then came to Bristol to pursue an MA in Wildlife Filmmaking at the University of West England. I’ve known for a long time that I wanted to get into filmmaking (since I was 12), growing up watching David Attenborough and living in the countryside greatly inspired me. However, there was no academic support in Spain for me to progress in this field, and so dreamed of getting into camerawork and research at the BBC Natural History Unit in Bristol; where a staggering 40% of the world’s wildlife documentaries are made. The Master’s course certainly helped me achieve this, and part of the MA involved making our own film, drawing upon all the skills we learned alongside it. Whilst my academic background was scientific, I had always loved being creative as a child; storyboarding, drawing and writing took me to the far-flung exotic and biodiverse places I saw on our television.
 Where I grew up in Southern Spain – the light and birdlife greatly inspired me.
Where I grew up in Southern Spain – the light and birdlife greatly inspired me.
Watching producers and cameramen/women filming behind the scenes sparked my interest; combining the best of both science and art worlds. Then when I was 13 my father bought me my first DSLR, and could finally capture the Bee-eater birds and Short-toed eagles that were always tantalizingly out of reach. Armed with my telephoto lens, I wanted to share my passion with others but knew that being self-taught wouldn’t be enough to cut it in this competitive industry.


I therefore had a lot of catching up to do at University; joining the Leeds Student Radio societies, television and photography clubs so that I could begin to create a portfolio and apply for the MA. This included a conservation YouTube Channel about local biodiversity, and two radio programs that I produced; Weekly Wildlife Watch and the Travel Talk Show. Whilst radio and LSTV taught me how to write, shoot and focus on story; PhotoSoc helped me to compose and learn about the fundamental principles of photography.


- Who has done what on the film?
As part of the MA requirement, I saw the entire project throughout each of the processes – from the initial idea as a researcher, producer/director, camerawoman, sound, editor, SFX/mixer, grader and now social media manager promoting the content online. It was certainly challenging juggling the different roles, but I loved learning and trying out a variety of methods, techniques and styles from both the drama and natural history world. Learning through mistakes is certainly the best way forward in all walks of life, and by having total creative and editorial control I feel I’ve made something close to my childhood memory and dream. It certainly has been an extraordinary experience that I will remember forever.

- And how did that utilise your individual skills?
With my camerawork, I attempted to adapt different styles of shooting as well as techniques to create a visual story that would suite a film festival audience. Such immersive filmmaking techniques include those seen on various BBC series capture the animal’s perspective to add an emotional level to the story, leading to a more powerful, captivating documentary. I attempted to capture privileged views of the lion in its environment, and learned a great deal about how different documentaries are made by analyzing the shots in different sequences.

I shot in high speed in attempt to create a sense of drama for the first and last sequence of A Lion’s Tale, as well as with the use of extreme close ups (in particular the ranger patrol) for an immersive feel. The storyboarding of key sequences proved to be invaluable during the shoot, as it allowed me to focus on what I wanted to achieve in terms of framing, direction, action, speed.

Equally, I took the camera off the tripod and onto a small inexpensive rig to allow for camera movement – and emphasizes the feeling and mood for a scene. The use of jibs, cranes and float cams are increasingly being used in natural history to create dynamic movement and a parallax between the scenes as seen in drama. The development of gimbals has now made this possible, although I had to resort to a very crude version of one in the end – my arms!

- So what’s the film about?
The Born Free story began with lions, and now 50 years later since the original film, A Lion’s Tale looks at the legacy that actress turn conservationist Virginia McKenna has left and the conflicts that lions and all wildlife face in Kenya. Set in the original heartland of the true father of lions, we journey to Meru National Park to see the Born Free team and Kenya wildlife service rangers on the front line of conflict and education. The world’s largest ivory burn is about to take place, as a symbol of Kenya’s determination to help all wildlife and stop the illegal trade. Will the next generation take up the challenge? Is there hope?

This film reveals how Kenya’s new generation of conservationists is looking out for all wildlife, including the elephants, which is witnessed at the world’s largest ivory burn event – a symbol of stopping all wildlife trade and helping humans and nature co-exist. Gaining access to this historical event was one of the greatest challenges and provided me with the opportunity to capture a unique moment in time. It has quickly been adopted in the media with several feature films and documentaries have highlighted the event (The Ivory Game, Hugh’s Ivory War). It was a truly unforgettable experience, which I self-shot and have now begun to edit into a separate film alongside this production. Filming beside my heroes, both in the conservation and camera world was one of the highlights of the shoot. The worlds press and filmmakers were gathered to document it, and it was truly a sobering sight to see the 150 tons of ivory go up in flames.





- Who wrote it and what inspired that?
The story of A Lion’s Tale began with my passion for lions and chance meeting with leading ape conservationist Ian Redmond.

This love of Africa and felines started during my childhood, when I was encapsulated by the true story of George and Joy Adamson. For me, the purpose of making A Lion’s Tale was to emotionally engage and raise awareness – focusing on one of the major issues not only concerning lions, but all wildlife in Kenya. The original Born Free story captured the emotions of millions during its release in 1956, a time when our relationship with the natural world and ‘wild’ animals was viewed negatively.



And so, whilst a large conservation movement was seemingly triggered with the momentum of a single film, it was the emotive driving force behind the true story about the real Adamson’s who released an orphaned lioness into the wild that led to actress Virginia McKenna to change her entire career and life plan – from actress to activist. I also felt it timely to produce with the upcoming ivory burn and Convention on the International Trade on Endangered Species meetings in Johannesburg. I didn’t want to write a set script; and by using the characters’ voices in the film I hope this has allowed audiences to connect with and care about the cause – not be lectured on it. The major theme of the film is hope – an emotion that all humans can relate to and a message that I believe everyone involved in the filmmaking and conservation industry can use as a device to inspire and drive change.
- And who produced it and pulled the project together?
I was the producer of the project and responsible for all the script-writing, scheduling, budgeting, interviewing, shooting, etc. which did take extensive planning. After conducting all the research, calling and making the contacts; getting out on location was thrilling but accounted for only 20% of the production! Logistically it was challenging, Meru National Park is not a well visited park like the Maasai Mara or Amboseli. I booked a direct flight from London Gatwick to Nairobi to go and film at the Born Free office based there, the ivory burn and then a small carrier plane into the heart of Meru for the lions.
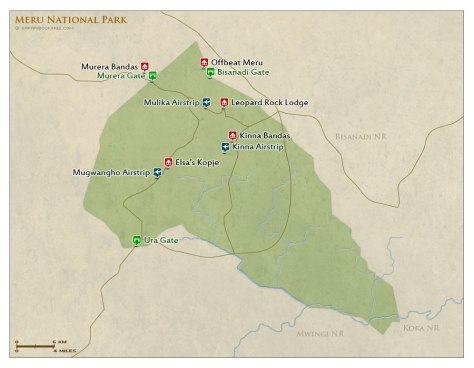

Upon descending Meru, all I could see was a small office – no runway as such but more of a dirt track – then suddenly a giraffe galloping away from us in attempt to avoid a collision! However, the Born Free team and Kenya Wildlife Service were remarkable, they helped make the shoot a success – driving me to all the locations within this most beautiful and underrepresented of parks. The ivory burn was undoubtedly the hardest to get permits for; but with a lot a patience and incredible support from the Born Free’s president (CEO) as well as one of my contributors, Will Travers, I was fortunate enough to be able to film at the historic event.


- Did you have much in the way of money to play with?
Not at all! I calculated a rough budget of £3000, as most of my negotiations brought the prices down and the park fees to film were waivered in return for me editing a separate version for them. This may seem ludicrous to most drama filmmakers, but in wildlife the budgets are far smaller and so this is where precision also plays a part throughout the production process. In total it cost £2600 for a 10 day shoot – this of course excludes all the pre-production and post costs as I was the one researching, filming, directing/producing, editing, grading, sound mixing; however the music was beautifully composed by MA student Richard Collins as part of his course. The facilities were provided by my university and the training in advance, but also a lot of practical reading and watching hours and hours of ‘How to…’ videos! Kit was also borrowed from the university and so most of the budget was spent on flights and accommodation. I did however set up a crowdfunding campaign and managed to raise half the funds to go; kind support from family, friends and strangers alike. IndieGoGo was the platform I used as it’s less risky if you don’t hit your top target.


- And what were your kit choices for this film?
Kit wise I used what was available at university; the Sony FS700 with the kit lens (18-200mm f/3.5-6.3), the 50mm f/1.8, 100mm Canon f/2.0 primes and the 100-400mm with the EF metabones adaptor. The telephoto was crucial for getting close to the action when it would have otherwise been too far and dangerous. This was especially the case with the lions! For sound; radio mics, Sennheiser 416 with the 522 mixer, and a Tascam for good measure – the latter was used to record atmos in the field. The wild sounds of Kenya truly are as vivid and vibrant as you would imagine. I loved recording the young group of school children who sang to us, it stirs up many joyful memories when played back.




The film was edited on Premiere Pro, Pro Tools, and graded in Da Vinci. In hindsight, I would have loved to have taken an DJI Ronin MX gimbal, FS7 and a Phantom 4 Pro drone for the aerials; but very grateful for the access to the kit we had, especially as students. You are only ever truly limited by your own imagination.
- Where does it sit alongside the rest of your portfolio of work?
In terms of technical difficulty, time scale, and aspiration to make – it’s right up there! As I am sure many of you reading this have experienced, we are our own worst critics. However, this is one project that I was excited and dare I say it, proud to have made. It’s been a life-long ambition to meet my heroine Virginia McKenna, and never thought I would get to experience the true Born Free story alongside the incredible people who keep that spirit of the Kenyan wilderness alive.

- So where are you with the film right now?
The film is now complete, however there are a few colour grading tweaks being done by a professional, as I now have the confidence to hit the bigger festivals! It was something that I lacked skill-wise, and could only grade and colour correct to a certain extent. I’m looking forward to the festivals and so far, it’s collected awards at six in the US, UK, India and Spain; winning recently at the Wild Film Fest in Falmouth. But more importantly, it’s been shared in the schools back in Meru where it was filmed, and where the real difference in changing attitudes towards these amazing animals can be made. They are the generation that can make all the difference.

- How is it looking at this stage in the game?
All finished! It took me 10 months in total, with my fellow course mates and I recently enjoyed a screening of our films at the Everyman theatre in Bristol. It was incredibly rewarding to see in on the big screen with family and friends, as well as some BBC staff who came to support us. A project that you are so involved in does, to a certain extent, take over your life for a while – but it’s such an incredible feeling to see the end creation. Although I always say that if I didn’t set myself a cut-off point, I’d be forever editing! I received a heartfelt letter from Virginia McKenna, my main character, about how much she enjoyed the film – and that was very special indeed as she had a huge influence on me when I was growing up.
- And how do you plan to promote it given that this is such a competitive marketplace?
I plan to promote it through a variety of social media platforms; Twitter, Instagram, Facebook, etc. and by connecting with influential Born Free supporters who can use their media presence to share and connect the film with the intended audience. Equally by continuing to enter festivals, I hope to promote it further worldwide. I also shot some 360 VR ‘behind the scenes’ clips to showcase on my website, especially during the ivory burn. Tapping into this market is key to reaching the younger audiences or those not necessarily interested in wildlife. During the run up to the film’s creation, I built up an online audience offering exclusive clips and images from the shoot as well as other stages of its development. It’s really important to engage with them and respond to what they have to say; as well as offer any advice. They also want their voice to be heard.
- Can you tell us about the other projects you’ve been working on?
 At the moment, I am employed at the BBC as a researcher with the digital team – one of the most innovative, creative and energetic group you’ll meet at the Natural History Unit! I’m loving every moment, most recently we released exclusive Snapchat stories for Planet Earth II in the US, and now I’m working on another digital project associated with the Blue Planet series. It’s incredibly exciting as you get to help out in various productions with different roles. Film-wise I do have a couple of personal project ideas in the pipeline, and I am very keen to shoot another short using the superb Panasonic GH5 – watch this space!
At the moment, I am employed at the BBC as a researcher with the digital team – one of the most innovative, creative and energetic group you’ll meet at the Natural History Unit! I’m loving every moment, most recently we released exclusive Snapchat stories for Planet Earth II in the US, and now I’m working on another digital project associated with the Blue Planet series. It’s incredibly exciting as you get to help out in various productions with different roles. Film-wise I do have a couple of personal project ideas in the pipeline, and I am very keen to shoot another short using the superb Panasonic GH5 – watch this space!

- How do they differ from this one?
They differ in that they are not located in Africa! I’ve always been incredibly drawn to the continent but now have storylines I’m researching in Australia and Japan, both having more of a cultural-human element to them. However, another short I’d love to shoot is more of a pure wildlife blue-chip style, and I now have access to better equipment and financing to facilitate the projects.
- What is your favourite genre and why?
I’m a little biased when saying I adore making natural history, but it’s something I live and breathe every second of the day. For centuries, humans have told stories to make sense about the world- illuminating behaviour, making order out of chaos and to create moral meaning. It’s the way we can comprehend and pass on information, with which we have the insatiable need for form and structure in the way we tell them. Natural History has been documented for thousands of years through visual, physical and audible means, whether through the primitive Stone Age depictions of a hunt through cave paintings, to the now pioneering ultra-high definition wildlife films. Nature is endlessly fascinating and beautiful, and as a curious person it’s something that I’m always passionate and keen to share with others. I am certain most of us have this desire to learn about the world around us. I do of course enjoy watching a great variety of programmes and films; adventure, comedy, sports, fact ent, animation and action! You can learn many lessons from different genres.


- Are there other genres that you’d like to tackle?
Natural History will always be my passion – but I do believe you can cross-pollinate genres and get interesting results. A superlative example the award-winning success Virunga directed by Orlando Von Einsidel; where wildlife documentary meets investigative journalism. It combines elements of “The seven key steps of story structure” as described by screenwriter John Truby, and regardless of it being a non-fiction film, Virguna also contains strong dramatic elements and a classical story structure of good versus evil. Battles depicted through the civil war and conflict between the park rangers and the oil companies are also tied in with the need to survive, along with the desire to exploit natural resources for profit. These all conjoin into a single cause and effect pathway through the combined use of a ‘run and gun’ shooting style and profound emotive pauses. It would be interesting to try a more daring, journalist approach with a conservation story – thrilling audiences by being immersed right in center of the action.
- So what is the filmmaking climate like in your neck of the woods?
Wildlife filmmaking is quite different to drama in my experience. Whilst we are adopting more cinematic techniques and technology using gimbals and aerials to create a parallax and dynamic edge; the set-up times, cast (!), budgets, and approach are quite different. I worked recently on a drama set as a camera assistant and found the whole thing fascinating; there certainly are no repeat takes when filming a wild animal in action! Equally, whilst in drama you have many specialized roles such as focus pullers, gaffers, and make up; wildlife crews are noticeably smaller as costs for location shoots would soon quickly rise – budgets are noticeably smaller. Most crews are a jack of all trades, and you learn quickly how to be as multi-skilled and useful to a team as possible. It’s the most incredibly rewarding and thrilling industry, and you never quite know what animals you will see and how they will behave. If you’re lucky, you can capture unique behavior that’s never been seen before.

- Are you at the stage of making any money from this as yet?
No, I decided that this project would not be for profit, despite my access to the ivory burn. I wanted this to be an educational and inspirational piece and shared far and wide; available to anyone with an interest in wildlife and Africa. Now that I know I can make a short film – next time may be different!
- So where do you see this filmmaking route taking you in the future?
I hope to follow in the footsteps of some of my filmmaking heroes; Sophie Darlington, Sue Gibson, Justine Evans – the best female camerawomen in our industry! But equally, I do want to pursue my passion for producing and continue to create compelling stories. I recently met the producers who worked on Planet Earth II who were incredibly inspiring- their work is truly in a league of its own. David Attenborough of course has been the greatest source of inspiration; and If I can make entertaining, emotive and compelling films that inspire others to want to make even the smallest of positive differences on our beautiful planet, then I’ll be a very happy earthling indeed.










.jpg)











































































